Futuristic implanted chip delivers osteoporosis drug in small clinical trial
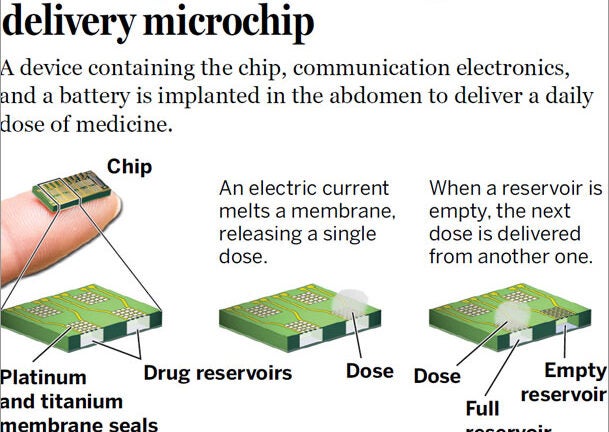
It sounds like science fiction: a doctor implants a device about the size of a domino just under the skin near a patient’s waistline. Over the course of weeks, tiny sealed wells on a chip embedded on the device open one by one, to release a potent drug on a schedule sent to it wirelessly.
But the futuristic scenario is real. Scientists reported today the first successful use of the novel technology in a small number of osteoporosis patients, 15 years after a MIT bioengineer was inspired by a television show about how computer chips are made.
“You could deliver many different medicines at once, a pharmacy on a chip,’’ said Robert Langer, the MIT bioengineer who led the work. “You could do … remote control delivery, kind of like Star Trek.’’
The technology opens the door to a tantalizing array of possibilities: devices that could be programmed to release a drug by a doctor from afar, or implants that could automatically sense when a diabetic person’s blood sugar levels were dangerously low and release a drug. But the device, being developed by a small Waltham company, MicroCHIPS Inc., is still far from changing how the medicine goes down.
Even now, it will take several years to develop and test the next generation of the technology before seeking regulatory approval. And the chip is not likely to change how all drugs are given. Ultimately, it will be limited by the types of drugs that can be usefully packaged on the tiny microchip.
“When I look at the drug delivery field, there’s never a one-size-fits-all,’’ said Langer, who has been a pioneer in inventing new ways to get medicines into the body.
The scientists chose to develop a device that administers an existing osteoporosis drug that stimulates bone formation. That drug, human paraythyroid hormone, is potent, and the daily injections patients now get, make compliance with the drug regimen a problem.
“I would say at least 25 percent of patients in whom I recommend this therapy are really stopped because of the need to inject,’’ said Dr. Felicia Cosman, an osteoporosis specialist at Helen Hayes Hospital in West Haverstraw, N.Y., who was not involved in the study. “I do think that any new delivery systems which avoid the need to inject daily are really warranted and may aid in patient adherence to therapy.’’
In the new study, published online in the journal Science Translational Medicine, eight women with osteoporosis were implanted with the devices through a minor surgical procedure. The chips were programmed to release doses of the drug included in the prototype device once daily over 20 days, and all but one of them functioned. The scientists were able to monitor the drug in the body, and found that it was active similarly to when it was injected and increased bone formation — although six of the patients received implants that carried fewer than 20 doses of the drug. The patients reported they would repeat the procedure to have the implant again.
Dr. Bess Dawson-Hughes, director of the Bone Metabolism Laboratory at the US Department of Agriculture’s Human Nutrition Research Center on Aging at Tufts University, said the results are exciting because of the potential to improve patients’ compliance. Since the severely-affected patients who need the drug are generally elderly, forgetfulness or bad vision can mean missed doses.
Robert Farra, president of MicroCHIPS, said the company is developing a next generation of the device that would have a full year — 365 doses — of the osteoporosis drug on it.
“The biggest areas for us will be diseases that have drugs that are very potent and require frequent injections,’’ Farra said, adding that the company is also interested in the potential to develop a device that could deliver a multiple sclerosis drug, and possibly some biotechnology drugs. He said the company has designed devices with sensors integrated, opening the possibility that a chip in a patient’s body could wirelessly connect with a doctor’s office to report data or release a drug when levels of a key molecule change in a patient.
Ian Sanderson, a senior analyst at Cowen and Co. who follows the drug delivery field, said the technology is interesting, especially because of the possibility of combining drug release with sensing. Ultimately, however, he said the chip would likely have fairly narrow applications because of the limitation of tiny wells that hold the drug.
“It’s got to be a very potent and very stable drug,’’ Sanderson said. He said that what would ultimately make the device a compelling way to deliver a drug would be a combination of effectiveness and economics.
Farra said that it costs about $1,000 per month for the injection pen now used to administer the drug. Though it is still years away from being available, he projected that the implanted device with a year’s supply of drug would be comparable in price, about $10,000 to $12,000.
Correction: A business story describing an implanted drug delivery device yesterday was unclear about why some of the patients received fewer than 20 doses of an osteoporosis drug. All the doses of the drug were released, but some patients received chips that carried fewer than 20 doses of the drug.







To comment, please create a screen name in your profile
To comment, please verify your email address
Conversation
This discussion has ended. Please join elsewhere on Boston.com