FDA approves women’s sex drive drug – but don’t call it ‘female Viagra’
That’s not what Viagra does.
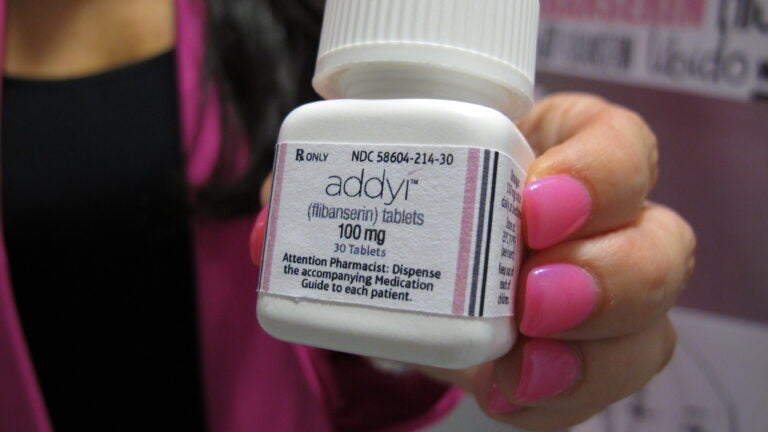
The Food and Drug Administration on Tuesday approved a prescription drug that increases women’s libido and sexual drive, the first drug to do so for either men or women.
The drug flibanserin, marketed as Addyi, has often been referred to in headlines and stories as the “female Viagra.’’ Like Viagra, the new pill is connected to sexual dysfunction, but that’s about where the similarities end.
Viagra, which treats erectile dysfunction in men, works by enhancing blood flow to the penis in order for men to maintain an erection. The drug takes 30-60 minutes to work, according to Viagra, which encourages people to “only take it when you need it.’’
Addyi, meanwhile, targets chemicals in the brain to increase sexual desire in women as a treatment for hypoactive sexual desire disorder, or a lack of sex drive. It should be taken daily for up to a month to be effective.
The decision to approve Addyi comes after an advisory panel voted in favor of its approval in June. The FDA had previously rejected the drug twice and cited its marginal effectiveness and side effects, including low blood pressure, fainting, and nausea, according to The New York Times.
The drug had been pushed by women’s health groups like Even The Score, which argued that the lack of a women’s sexual drug was evidence of a gender disparity that needed to be corrected.
Gallery: A parent’s guide to important vaccines.
[bdc-gallery id=”114778″]







To comment, please create a screen name in your profile
To comment, please verify your email address
Conversation
This discussion has ended. Please join elsewhere on Boston.com